 
In the case of C14, it has two additional neutrons compared to the standard carbon 12 atom. An isotope is an atom with an abnormal number of neutrons in its nucleus. Knowing the uses of the technique, however, does not answer the question, “What is radiocarbon dating?” Radiocarbon dating is a technique that estimates the exact age of organic materials based on the amount of C14 isotope present in the sample. Once a relationship has been established, however, it is possible to estimate the date of a wide range of archaeological discoveries based on the dating of the sample, including the date of a building’s destruction, the period during which a site inhabited, and more. Unfortunately, this is rarely a straightforward process and can sometimes introduce further errors into establishing a site or layer’s absolute date (discussed below). Beyond being able to date the sampled material to a specific date, the chronological information can then be used help date surrounding architectural features and archaeological finds. However, this is not where the story ends. These tests return an estimate of the age of the sample within a margin of error. Then the sample is sent to a specialized lab that can run the appropriate tests. Once organic materials have been identified, archaeologists must quickly place the find in an appropriate container to reduce the chances of contamination. This could be bones, charcoal, woolen textiles, or even ancient seeds. The most powerful of these is radiocarbon dating, but what is radiocarbon dating and how do archaeologists use it?įor archaeologists to properly date something using radiocarbon dating, they must first discover something made from or containing an organic material. Unfortunately, these sorts of textual clues are exceedingly rare in antiquity, and instead, archaeologists turn to more scientific techniques. An obvious example is an ancient text that gives the specific year in which a building was constructed. The second type of dating method is absolute dating. However, as the name implies, these methods only provide a date relative to other artifacts or sources of data and are, therefore, rarely able to provide archaeologists with specific chronological information. The first are relative dating methods such as stratigraphy, typology, and even linguistic analysis of texts. 
Conversely, burning fossil fuels since 1900 has steadily lowered the amount of radiocarbon, because coal, oil and gas are all formed from plants and animals that died millions of years ago and so have essentially no radiocarbon left.There are two main methods that archaeologists use to date artifacts and levels (or strata) from archaeological sites. In the 1950s and 1960s, nuclear weapons tests briefly doubled the levels of radiocarbon in the atmosphere and radiocarbon dating needs to account for this to remain accurate. Dating anything older than 50,000 years is very difficult since so little radiocarbon remains after that long, but some measurements of samples up to 75,000 years old have been made. Radiocarbon dating is believed to be accurate to within a few decades or centuries, with lower accuracy the older the sample. 
The older the sample, the less radiocarbon is still present. Carbon-14 is heavier, so is deflected less. This uses an electric field to accelerate carbon ions past a magnet that deflects their trajectory. Scientists can measure the ratio of carbon-14 to the stable isotopes carbon-12 and carbon-13 with an accelerator mass spectrometer (AMS). When a plant or animal dies, it stops absorbing new carbon, so the proportion of radiocarbon in their body gradually drops due to this radioactive decay. The half-life of carbon-14 (the period of time after which half of a given sample will have decayed) is about 5,730 years. Radiocarbon is unstable, and slowly decays back to nitrogen. Herbivores eat the plants, and carnivores eat the animals, and so the radiocarbon spreads through the food chain, eventually reaching even the deepest oceans. Plants absorb radiocarbon, along with normal carbon dioxide, during photosynthesis and incorporate it into their tissues. About 1.1 per cent of the carbon atoms on Earth are carbon-14. This quickly oxidises to create mildly radioactive carbon dioxide. High-energy cosmic rays, in the form of neutrons, strike nitrogen atoms in the upper atmosphere and convert them to the radioactive isotope of carbon, which is carbon-14 (also known as radiocarbon). By measuring the amount of carbon-14 remaining in organic materials, radiocarbon dating can determine the age of ancient human remains, fossils, archaeological artefacts, and even historical documents with remarkable accuracy. Radiocarbon dating is used by scientists – including archaeologists and geologists – to uncover the mysteries of our planet’s past. 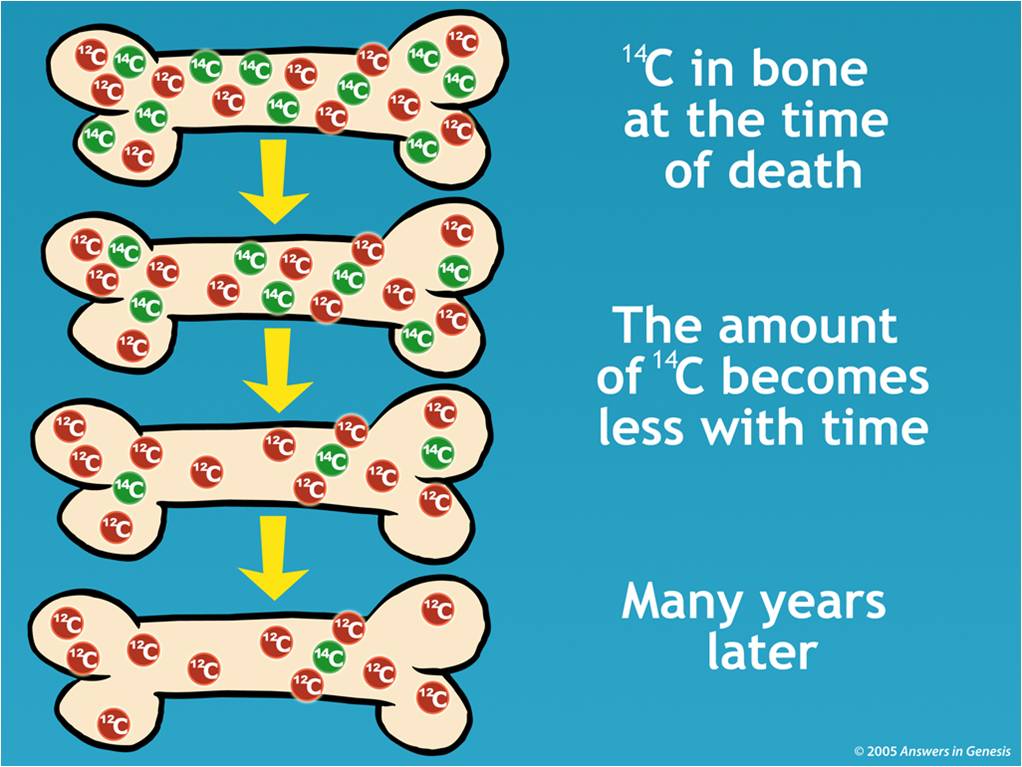
Carbon dating: How scientists hack radiation to age ancient artefacts 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
